Question
In: Chemistry
Draw the structure of the aromatic product from the following reaction
Draw the structure of the aromatic product from the following reaction
Solutions
Expert Solution
Concepts and reason
An acid anhydride reacts with amines to form an amide bond. Amide bond consists of CONH group, in which \(\mathrm{NH}\) group acts as a nucleophile and \(\mathrm{CO}\) group acts as an electrophile.
Fundamentals
Acid anhydrides are the compounds that are produced by removing water molecules from the acid and these act as nucleophiles. When an acid anhydride reacts with an amine, it forms a peptide bond.
The given substrate is shown below.
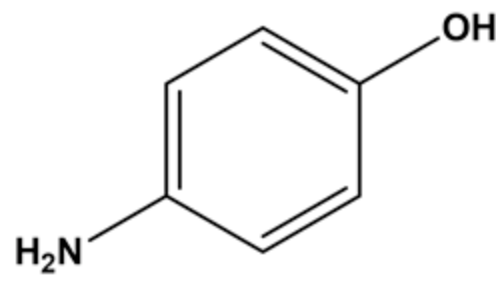
The given substrate consists of two functional groups namely \(\mathrm{OH}\) and \(\mathrm{NH}_{2}\). The amine group \(\left(\mathrm{NH}_{2}\right)\) is more nucleophilic than \(\mathrm{OH}\) since the electron density around \(\mathrm{NH}_{2}\) is more than \(\mathrm{OH}\).
The reaction can be written as follows.
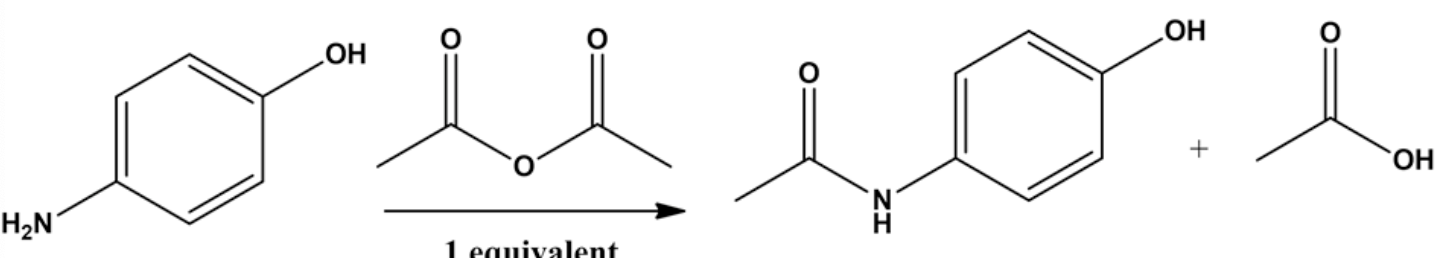
The structure of the aromatic product is

The amine group is more nucleophilic than \(\mathrm{OH}\) group. Hydrogen can be removed from amine \(\left(\mathrm{NH}_{2}\right)\) group thereby electrophilic attack of anhydride takes place resulting in the formation of the peptide bond.
The structure of the aromatic product is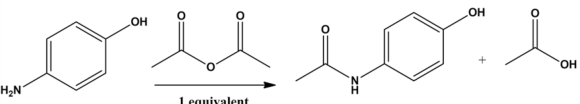
Related Solutions
Nitration of an aromatic ring involves an electrophilicsubstitution reaction. Draw the structure of the electrophile...
Draw the structure of the organic product of each reaction in the following two-step synthesis.
Propose a structure for an aromatic hydrocarbon, C9H12, that can form only one C9H11Br product on...
draw a mechanism that explains how the nitro-substituted aromatic products observed in your reaction were formed
Draw the reaction diagram from starting material to product of your specific reagent. This is for...
Draw the organic product for the following reaction. Omit any inorganic byproducts or ions.
What are the electrophile and nucleophilic in the electrophilic aromatic substitution reaction.
Draw the product of the oxidation of the following aldehyde. Include all hydrogen atoms in your structure.
Draw the major organic product of the reaction shown below.Draw the major organic product of...
Draw the organic product (if any) expected from the following reaction: (include all hydrogen atoms) CH3CH2CH2OH + K2Cr2O7(aq) H2SO4
- You have looked at the current financial statements for Reigle Homes, Co. The company has an...
- What is the circuit diagram of password based door lock system project by using interface 8088...
- java programming You will be given two interfaces and two abstract classes, FileTextReader, FileTextWriter, AbstractFileMonitor, and...
- Why is it significant that arginine vasopressin is expressed in squirrel monkeys and the amino acid...
- In the sigma-model of spontaneous symmetry breaking, we have degenerate vacuum states. But if we don't...
- Calculate the Kovats retention index for an unknown using the following retention times: 1.8 min for...
- the following selected financial statement information is for Stevens Company December 31 2017 2016 Changes in...
 Dr. OWL answered 5 years ago
Dr. OWL answered 5 years ago