Question
In: Chemistry
N-Methyl-2-pyrrolidone is an aprotic solvent used in many industrial processes. Draw the structure of the product formed when it is heated with strong aqueous acid 6MHCI, H20 and heat.
N-Methyl-2-pyrrolidone is an aprotic solvent used in many industrial processes. Draw the structure of the product formed when it is heated with strong aqueous acid 6MHCI, H20 and heat.
Solutions
Expert Solution
Concepts and reason
This problem is based on the concept of the reaction of \(\mathrm{N}\) -methyl- 2 -pyrrolidone. N-methyl-2-pyrrolidone is an organic compound that consists of a five-membered lactam. It is called NMP as short. It is a beneficial compound that is used to recover hydrocarbons generated during the processing of petrochemicals.
Fundamentals
N-methyl-2-pyrrolidone is a very reactive compound. It reacts in an acidic medium to form a variety of products. Some reactions involve the ring-opening step.
The structure of N-methyl-2-pyrrolidone is given below:
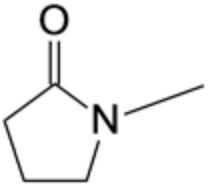
The compound has a five-membered ring, which consists of four carbon atoms and one nitrogen atom. The ring has one oxygen attached to it via the double bond, and one methyl group is attached to the nitrogen atom.
The chemical reaction involved is as follows:
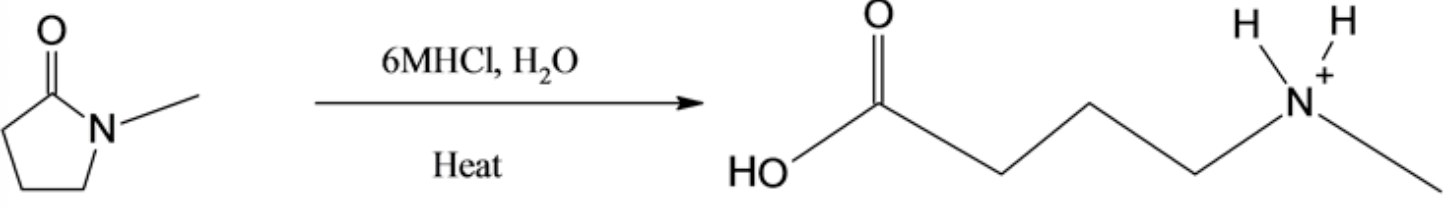
The structure of the product is given below:
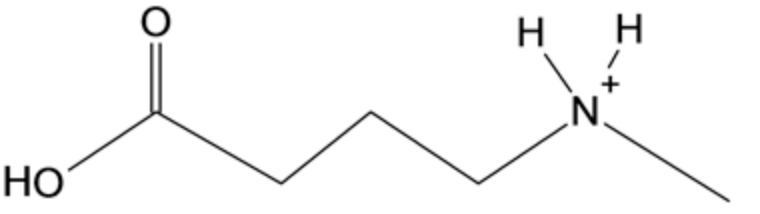
N-methyl-2-pyrrolidone on reaction with strong aqueous acidic medium followed by the treatment of heat results in the opening of the ring and forms a compound having a carboxylic group at one end and protonated nitrogen atom which has one methyl group attached to it, at another end.
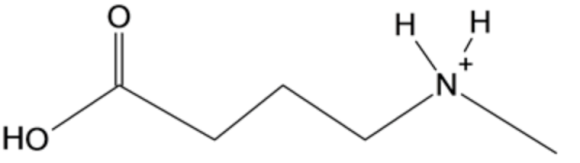
The structure of the product is given below:
Related Solutions
Butanone, when treated with methyl magnesium bromide and thendilute aqueous acid, yields 2-methyl-2-butanol.a) Please...
draw full sn1 mechanism for a reaction that includes 2-methyl-2-propanol, n-pentanol and hydrochloric acid as the...
1) Draw the structure of the product formed by addition of vitamin B1 and benzaldehyde. 2)...
1) Draw the structure of the product formed by addition of vitamin B1 and benzaldehyde. 2)...
1)When optically active (R)-2- methyl cyclohexanone is treated with aqueous acid , its optical activity is...
- A student has two lenses, one of focal length 5.5 cm and the other with focal...
- Show and explain each step (neatly in complete sentences), indicate units, and specify the correct number...
- Amazon and Google have implemented cloud-based DBMS, what are some pros and cons? What are some...
- Terminology: 1.Thickness of internal coating of an analytical column (meaning, the purpose and the range of...
- im learning how to code and i hear what is important are data structures and algorithms....
- You have looked at the current financial statements for Reigle Homes, Co. The company has an...
- What is the circuit diagram of password based door lock system project by using interface 8088...
 Dr. OWL answered 5 years ago
Dr. OWL answered 5 years ago