Question
In: Chemistry
Draw the organic products formed in the following reaction.
Draw the organic products formed in the following reaction.
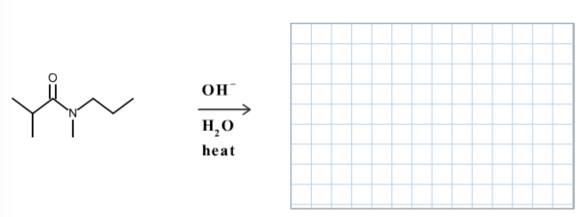
Solutions
Expert Solution
Concepts and reason
Amide hydrolysis is a process where an amide is hydrolyzed and forms a carboxylate ion in the presence of a base. In the presence of an acid, amide hydrolysis ends up with carboxylic acid and an ammonia derivative. Amide hydrolysis follows the mechanism of ester hydrolysis under basic conditions.
Fundamentals
- Amide hydrolysis under basic media produces the carboxylate ion and ammonia derivative. First, the base attacks the positively charged carbon atom in the carbonyl group of amide and forms oxy anion. Then, the oxy anion stabilizes to form carboxylic acid by giving the amine derivative. Under basic conditions, the carboxylic acid formed loses the proton to form a carboxylate ion.
- General amide hydrolysis under basic conditions
Given amide is shown below.
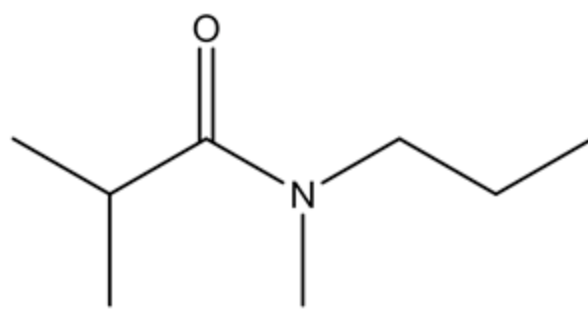
Base attack on the carbonyl group of the amide
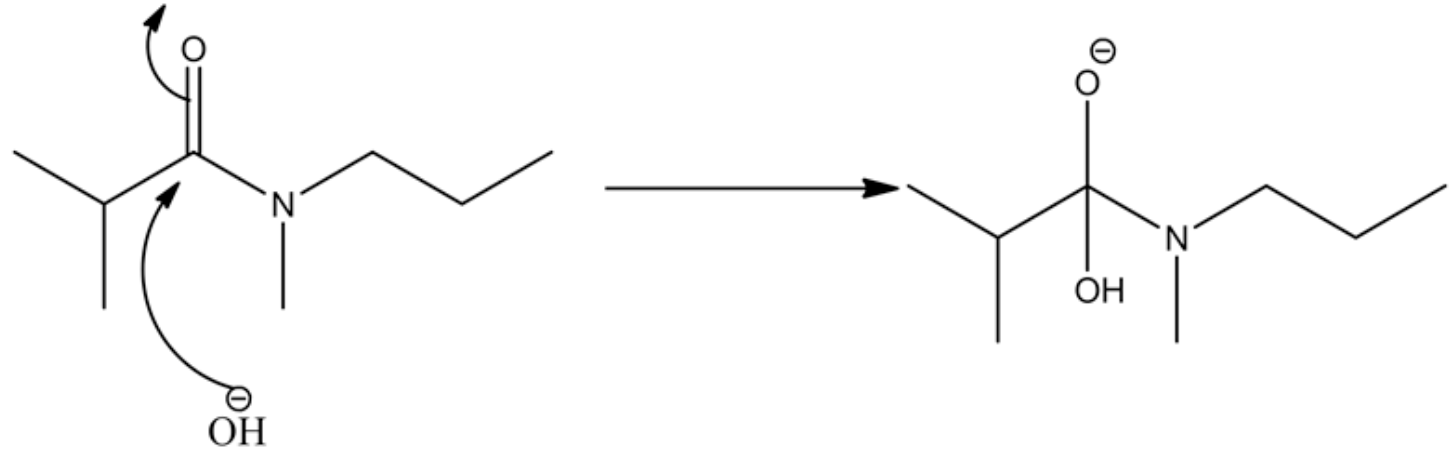
Oxy anion is formed during the attack of the base on a carbonyl group of amide. The base attacks the partially positive carbon atom of the carbonyl group and produces the oxyanion, where the negative charge lies on the more electronegative oxygen atom.
Oxyanion
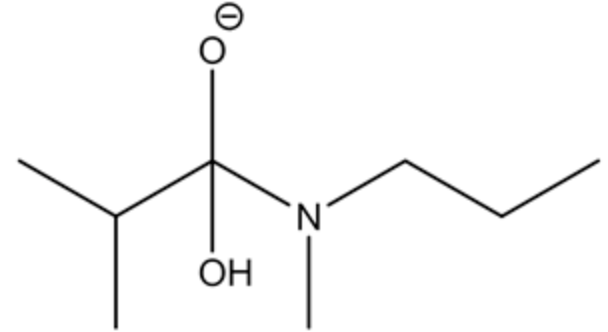
Stabilization of oxyanion
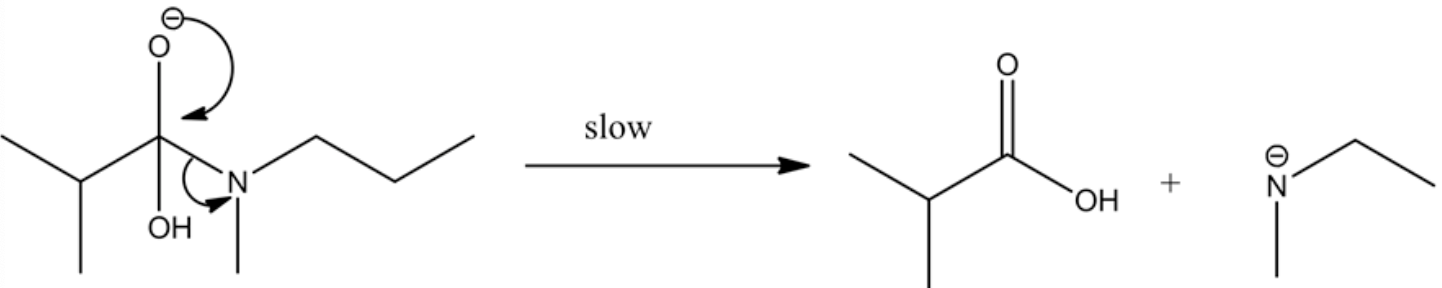
Stabilization of oxyanion to carboxylic acid is a slow step process, whereas carboxylic acid to carboxylate conversion in the presence of a base is very rapid.

The complete reaction
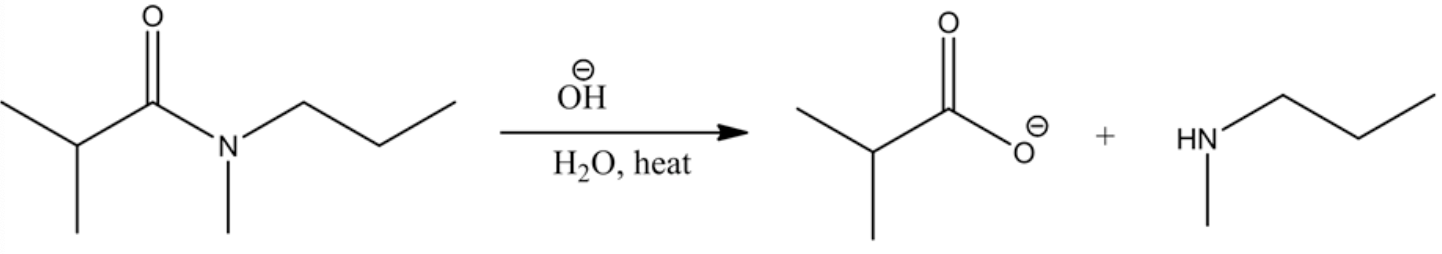
Stabilization of oxyanion to carboxylic acid is a slow step process while carboxylic acid to carboxylate conversion in the presence of a base is very rapid. An amide ion abstracts the acidic proton from the carboxylic acid very rapidly and forms a carboxylate ion. The carboxylate ion is highly stabilized through resonance.
The complete reaction
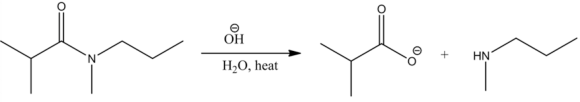
Related Solutions
For the following SN2 reaction, draw the organic and inorganic products of the reaction, and identify...
Draw the structures of the organic products in each reaction of the following two-step synthesis.
For the following SN2 reaction, draw the organic and inorganic products of the reaction, and identify the nucleophile, substrate, and leaving group.
For the following SN2 reaction, draw the organic and inorganic products of the reaction, and identify the nucleophile, substrate, and leaving group.
Draw the major organic product(s) for the following reaction. Multiple products may be drawn in one...
Draw the organic products formed when allylic alcohol is treated with each reagent a. H2 +...
Draw the major organic product of the reaction shown below.Draw the major organic product of...
Draw the organic product for the following reaction. Omit any inorganic byproducts or ions.
Draw the structure of the organic product of each reaction in the following two-step synthesis.
draw a mechanism that explains how the nitro-substituted aromatic products observed in your reaction were formed
- state a complete sales contract of any product including bill of lading in international trafe
- Consider the following assets available for investment: 1. A stock index fund 2. A corporate bond...
- Program a calculator or computer to use Euler's method to compute y(1), where y(x) is the...
- c++ 1 city.h #pragma once #ifndef CITY_H #define CITY_H #include<string> #include<iostream> #include<utility> using namespace std; class...
- 1. Complete the balance sheet for the business for 2016 and 2017. 2. In which year...
- 1. What is oxidation? How do you know if a molecule/atom has undergone oxidation. 2. Describe the...
- write a simple python program that takes in 6-50 random numbers the user types in and...
 Dr. OWL answered 5 years ago
Dr. OWL answered 5 years ago