Question
In: Chemistry
When 0.601 g of biphenyl (C12H10) undergoes combustion in a bomb calorimeter, the temperature rises from...
When 0.601 g of biphenyl (C12H10) undergoes combustion in a bomb calorimeter, the temperature rises from 26.1 ∘C to 30.3 ∘C.
Find ΔErxn for the combustion of biphenyl in kJ/mol biphenyl. The heat capacity of the bomb calorimeter, determined in a separate experiment, is 5.86 kJ/∘C.
Solutions
Expert Solution
Given
Mass of biphenyl = 0.601 g
Heat capacity of the bomb calorimeter = 5.86 k J / 0 C
Change in temperature = 30.3 0 C - 26.1 0 C = 4.2 0 C
Molar mass of biphenyl ( C12H10 ) = ( 12 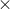 12.01 ) + ( 10
12.01 ) + ( 10 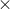 1.0079 ) = 154.20 g / mol
1.0079 ) = 154.20 g / mol
We have relation, 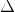 E = Heat capacity of the calorimeter
E = Heat capacity of the calorimeter 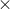 Change in temperature
Change in temperature 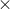 ( Molar mass / Mass )
( Molar mass / Mass )
 E
= 5.86 k J / 0 C
E
= 5.86 k J / 0 C  4.2 0 C
4.2 0 C 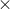 ( 154.20 g / mol / 0.601 g )
( 154.20 g / mol / 0.601 g )
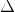 E
= 5.86 k J / 0 C
E
= 5.86 k J / 0 C 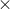 4.2 0 C
4.2 0 C 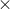 256.57 mol -1
256.57 mol -1
 E
= 6314.7 k J / mol
E
= 6314.7 k J / mol
ANSWER :  E = - 6.31
E = - 6.31  10 3 k J / mol
10 3 k J / mol
Related Solutions
When 0.595 g of biphenyl (C12H10) undergoes combustion in a bomb calorimeter, the temperature rises from...
A 1.000 g-sample of a compound undergoes combustion in a bomb calorimeter. The temperature of the...
A. When a 0.235-g sample of benzoic acid is combusted in a bomb calorimeter, the temperature...
Q) The combustion of 3.07 g of hydrogen in a bomb calorimeter with a heat capacity...
In an adiabatic, constant-volume bomb calorimeter, a combustion of 0.7362g of naphthalene (C10H8) caused the temperature...
A sample of K(s) of mass 2.720 g undergoes combustion in a constant volume calorimeter at...
When a 3.125 g sample of ammonium nitrate decomposes in a bomb calorimeter with a heat...
41. When 0.500 g of cyclohexane, C6H12, is combusted in a bomb calorimeter that has a...
a 5.00 g sample of TNT (C7H5N2O6) is burned in a bomb calorimeter with a heat...
A 21.8 g sample of ethanol (C2H5OH, 46.07 g/mol) is burned in a bomb calorimeter, according...
- Two common ways for a chemist to increase the yield of a reactiom are described below....
- Yahtzee Simulation. Must be written in Python These are the specific instructions: "Write a function called...
- League Competition: Suppose the demand for soccer teams in the US is given by: P(Q) =...
- two happy astronaunts, each having a mass of 75 kg are connected by a rod length...
- QUESTION 1 Compute the typical thermal energy of a water molecule at normal conditions: (kB T)...
- i used this coding for select image from database but the image did not retrieve from...
- Experiment A B C intial rate of formation of D (M/min) 1 2.0 2.0 2.0 2.0...
 queen_honey_blossom answered 2 months ago
queen_honey_blossom answered 2 months ago