Question
In: Biology
The following reaction is a part of the TCA cycle: succinate + FAD ⇄ fumarate +...
The following reaction is a part of the TCA cycle:
succinate + FAD ⇄ fumarate + FADH2
3) The E°’ for the reduction of FAD is -0.22 V and the E°’ for the reduction of fumarate is 0.03 V. Calculate ΔE°’ for the full reaction given above. Show your work. Be mindful of units. (1 pt)
4) Use your answer from part (3) to calculate ΔG°’ for the full forward reaction given above. Use F = 96,500 J V-1 mol-1. Show your work. Be mindful of units. (1 pt)
5) The ΔG°’ for the forward reaction is positive, and yet we know the TCA cycle is able to proceed in the cell. Is this a contradiction of thermodynamic principles? In two to three sentences, briefly explain your reasoning. (2 pts)
6) Let’s look at the same reaction, but under a new set of conditions that more closely match intracellular concentrations: [FADH2] / [FAD] = 10 [succinate] = 1.7 mM [fumarate] = 100 µM Derive an equation to calculate ΔE from ΔE°’, n, R, T, F, and the concentrations provided above. n = number of electrons Use R = 8.314 J mol-1 K-1, T = 298 K, and F = 96,500 J V-1 mol -1. Simplify the equation as much as you can before calculating ΔE. Show your work. Be mindful of units. Only round at the very end and write your answer out to four decimal places. (2 pts)
I just need help on parts 5 and 6 please explain
Solutions
Expert Solution
the problem is based on the concepts of thermodynamics.
5)
in the cells the hydrolysis of ATP releases a large amount of free
energy. that's why ATP bonds are also called the high energy bonds.
now the TCA cycle which is also called as Krebs cycle has the
negative Gibbs free energy though it proceeds in the forward
direction. Gibbs free energy is defined as the useful energy which
is used to determine whether the reaction will occur spontaneously
or not. if the Gibbs free energy is negative, it means that the
reaction will occur spontaneously and it it is positive, it means
that it need energy from external source for the reaction to
happen. so, for TCA cycle Gibbs free energy is positive but it
still occurs , this not against the thermodynamics as the ATP
energy will provide external energy for the cycle to occur.
6
 )
)
we have to derive the relation between E ,E0 , n , F , R, T , which is called Nernst equation.
let us take the reaction,
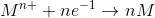
here, M ion reduces to M by combining it with n electrons.
so, the work done in reduction will be given as,
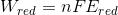
here ,
F is faradays's constant = 96487 coulomb
n = number of electrons.
so,
work required for the reduction and Gibbs free energy is related as,
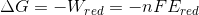
now at the standard temperature of 298 K , Gibbs free energy will be given as ,
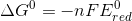
Ered = reduction potential.
so,
for a reversible equilibrium reaction, vant Hoff isotherm is given as,
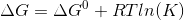
here
K = equilibrium constant.
hence for the given reaction it is given as,
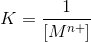
we substitute the values we get,
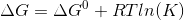
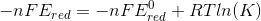
so we get
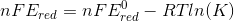
we divide the relation with nF on both side we get,
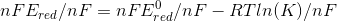
we get
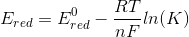
so we get,
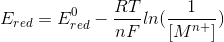 ................ [ans.]
................ [ans.]
hence we have derived the relation between the E, E0 , n, F, R, T
now for T = 298 K
R = 8.314 J/mol.K
F = 96500 C.
so, we get
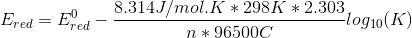
we get
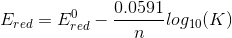 ...........
[ans.]
...........
[ans.]
now in this problem we are given that ,
K = [FADH2] / [FAD] = 10 [succinate] = 1.7 mM [fumarate] = 100 μM
so,
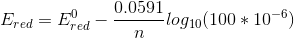
we get
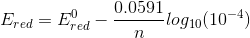
we get
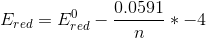
we get
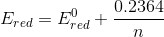 ............[ans.]
............[ans.]
so, if we know the n, and E0 values we can calculate the E value.
Related Solutions
The enzyme succinate dehydrogenase acts on succinate and convert it to fumarate tn the TCA cycle...
The oxidation of Succinate to Fumarate produces: a. the same amount ATP as the oxidation of...
The pyruvate dehydrogenase complex catalyzes the reaction pyruvate → acetyl-CoA (Reaction 0 in the TCA cycle)....
Succinate and fumarate have been shown to accumulate in some cancer cells. Dysregulation of which two...
For each of the following TCA Cycle enzymes, list the name of the enzyme in the...
Explain the benefit of the TCA cycle in detail.
Which of the following involve glycolysis, the TCA cycle, and electron transport chain? A. aerobic respiration,...
Describe how the fumarate produced by the purine nucleotide cycle could be catabolized to CO2.
39) Most of the enzymes for the TCA cycle are located ________ while most of the...
3. A class of mutations in TCA cycle enzymes result in a “hypermethylator phenotype” in a...
- You place your lunch leftovers in the refrigerator. Suppose the refrigerator needs to remove 2.0090E+4 J...
- A) What should his speed be at the top of the ramp to make it to...
- Please write a very brief reflection about "it's in her eyes, an unspoken pain in laboring...
- **SAP Project Manager What they do What tasks they perform What their career path could be...
- Assume you are 25 and earn $31,000 per year, never expect to receive a raise, and...
- A lidar (laser radar) gun is an alternative to the standard radar gun that uses the...
- You can buy a car that is advertised for $24,600 on the following terms: (a) pay...
 gladiator answered 2 months ago
gladiator answered 2 months ago