Question
In: Chemistry
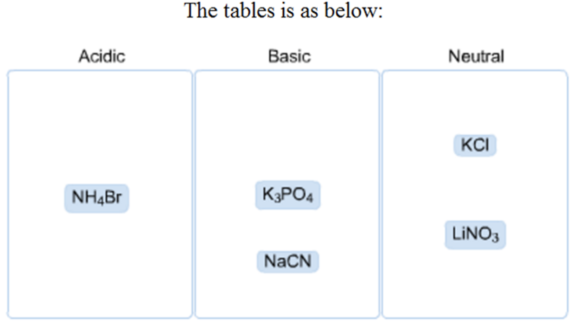
Classify these salts as acidic, basic, or neutral.
Classify these salts as acidic, basic, or neutral.
Solutions
Expert Solution
Compound: KCl
It has a cation of \(\mathrm{K}^{+}\) and \(\mathrm{a}\) anion of \(\mathrm{Cl}^{-}\)
The cation \(\mathrm{K}^{+}\) is from a strong base and the anion \(\mathrm{Cl}\) - is from strong acid.
So, it is neutral.
Compound : \(\mathrm{LiNO}_{3}\)
It has a cation of \(L i^{+}\) and an anion of \(\mathrm{NO}_{3}\)
The cation \(L i^{+}\) is from a strong base, and the anion \(\mathrm{NO}_{3}\) is from strong acid.
So, it is neutral
Compound : \(\mathrm{NH}_{4} \mathrm{Br}\)
It has a cation of \(\mathrm{NH}_{4}^{+}\) and a anion of \(\mathrm{Br}\)
The cation \(\mathrm{NH}_{4}^{+}\) is from a weak base, and the anion \(\mathrm{Br}^{-}\) is from strong acid.
So, it is acidic
Compound : \(\mathrm{K}_{3} \mathrm{PO}_{4}\)
It has a cation of \(\mathrm{K}^{+}\) and an anion of \(\mathrm{PO}_{4}\)
The cation \(K^{+}\) is from a strong base, and the anion \(\mathrm{PO}_{4}\) is from a weak acid.
So, it is basic
Compound : \(\mathrm{NaCN}\)
It has a cation of \(\mathrm{Na}^{+}\) and a anion of \(\mathrm{CN}^{-}\)
The cation \(\mathrm{Na}^{+}\) is from a strong base and the anion \(\mathrm{CN}\) ' is from a weak acid.
So, it is basic
Related Solutions
Complete the table below and classify the following salts (acidic, basic or neutral) and their pH...
Classify these amino acids as acidic, basic, neutral polar, or neutral nonpolar.
Determine if the following salts will be acidic, basic, or neutral. For each salt show the...
Classify these salts as acidic, basic, or neutral.NH4ClO4 NaF LiNO3 KCI K2CO3
Indicate if the aqueous solutions of the following salts would be acidic, basic, neutral, or “need...
Indicate if the aqueous solutions of the following salts would be acidic, basic, neutral, or “need...
Identify solutions of the following salts as acidic, basic or neutral. Identify solutions of the following...
State whether 0.1 M solutions of each of the following salts are acidic, basic, or neutral....
Determine whether aqueous solutions of the salts below are acidic (a), basic (b) or neutral (n)....
Predict whether aqueous solutions of the salts will be acidic, basic or neutral. Please explain why,...
- Show and explain each step (neatly in complete sentences), indicate units, and specify the correct number...
- Amazon and Google have implemented cloud-based DBMS, what are some pros and cons? What are some...
- Terminology: 1.Thickness of internal coating of an analytical column (meaning, the purpose and the range of...
- im learning how to code and i hear what is important are data structures and algorithms....
- You have looked at the current financial statements for Reigle Homes, Co. The company has an...
- What is the circuit diagram of password based door lock system project by using interface 8088...
- java programming You will be given two interfaces and two abstract classes, FileTextReader, FileTextWriter, AbstractFileMonitor, and...
 Dr. OWL answered 5 years ago
Dr. OWL answered 5 years ago