Question
In: Chemistry
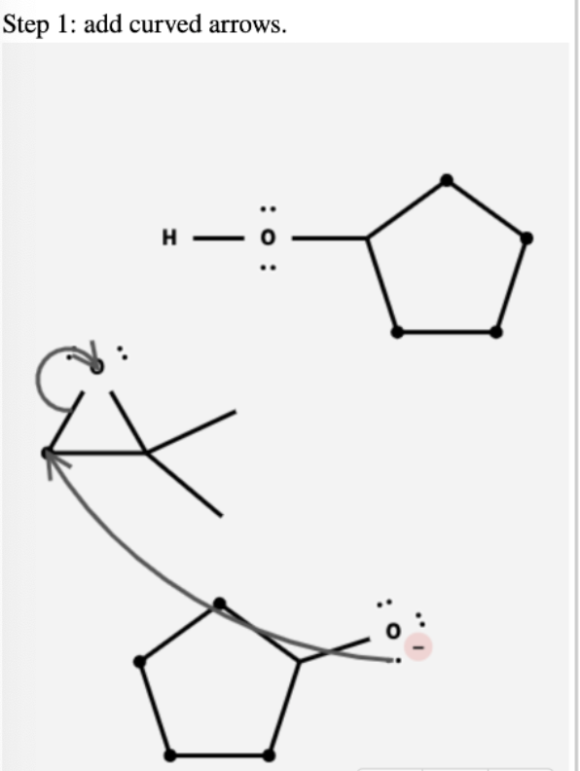
Complete the mechanism for the base-catalyzed opening of the epoxide by adding any missing atoms, bonds, charges, nonbonding electron pairs, and curved arrows.
Complete the mechanism for the base-catalyzed opening of the epoxide by adding any missing atoms, bonds, charges, nonbonding electron pairs, and curved arrows.
Solutions
Expert Solution
Opening of unsymmetrical epoxide is a regioselective reaction. The opening of unsymmetrical epoxide is depended upon the reaction medium i.e. either in acidic medium or basic medium.
In an acidic medium, the opening of epoxide takes place at the sterically more crowded side because the first step is the protonation of epoxide oxygen. Then it opens at which side depends upon the stability of carbocation formed. Always the sterically more crowded side opens.
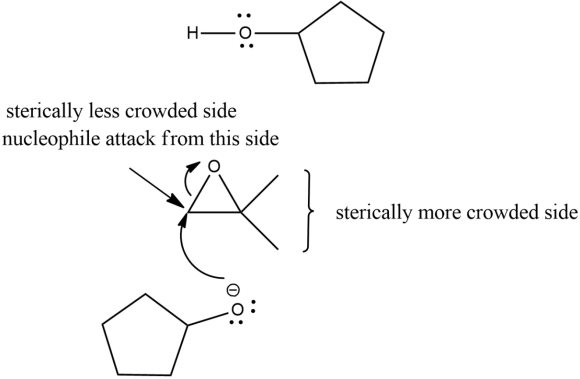
In a basic medium, the opening of epoxide is always at a sterically less crowded side takes place. Because the protonation at epoxide oxygen does not take place.
Step 1: In this step, the nucleophile opens the epoxide from the sterically less crowded side.
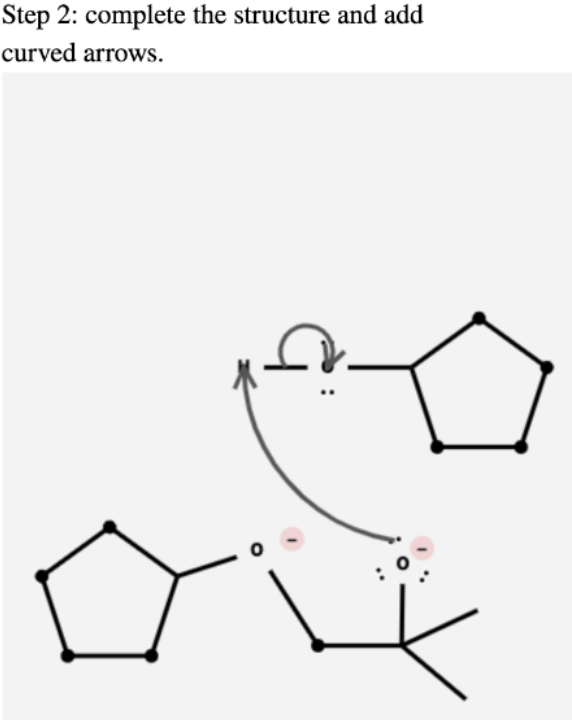
Step 2: In this step the protonation of oxygen takes place. The alcohols are good proton donors. So, the oxygen abstracts hydrogen from the cyclopentanone.
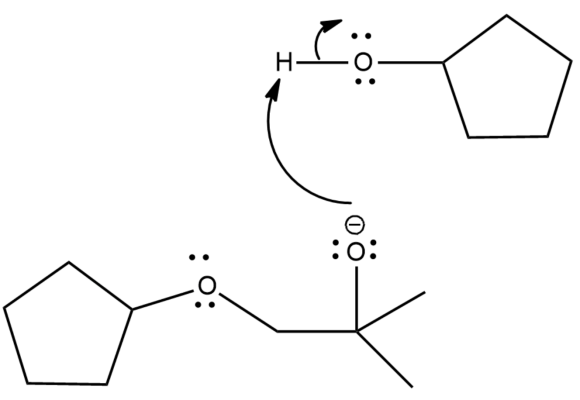
Step 3: In this step, the epoxide is converted into alcohol.
Related Solutions
Use curved arrows to predict the complete mechanism of formation of the imine shown below.
Complete the mechanism of the following Diels-Alder reaction by drawing the curved arrows for the concerted...
Draw the Lewis structure of H2O. Include any nonbonding electron pairs. Draw the molecule by placing...
- You have looked at the current financial statements for Reigle Homes, Co. The company has an...
- What is the circuit diagram of password based door lock system project by using interface 8088...
- java programming You will be given two interfaces and two abstract classes, FileTextReader, FileTextWriter, AbstractFileMonitor, and...
- Why is it significant that arginine vasopressin is expressed in squirrel monkeys and the amino acid...
- In the sigma-model of spontaneous symmetry breaking, we have degenerate vacuum states. But if we don't...
- Calculate the Kovats retention index for an unknown using the following retention times: 1.8 min for...
- the following selected financial statement information is for Stevens Company December 31 2017 2016 Changes in...
 Dr. OWL answered 5 years ago
Dr. OWL answered 5 years ago