Question
In: Other
A 5,000 mol/h fuel is fed to a furnace and burned with excess air. The fuel...
A 5,000 mol/h fuel is fed to a furnace and burned with excess air. The fuel contains 80.0 mole % carbon (C), 15.0 mole % hydrogen (H) and the remainder sulphur (S). The ratio of feed fuel to feed air is 1:7. The selectivity of carbon dioxide (CO2) to carbon monoxide (CO) production is 12. The conversion of fuel in this process is only 80%. One of the components in the flue gas is sulphur dioxide (SO2).
(a) Write all equations that involved in this
combustion process.
(b) Draw and label the process completely.
(c) Using Atomic Species Balance method, determine the molar flow
rates (mol/h) of all components in the product. stream.
(d) Determine the percentage of excess air in this process.
Solutions
Expert Solution
Fuel flowrate = 5000 mol/h
The fuel composition
80 mol% C
15 mol% H
5 mol% S
The ratio of fuel fed to air = 1: 7
Molar flowrate of air = 7(5000) = 35000 mol/h
A)
The equations involving are
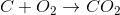
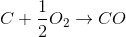
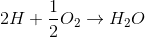
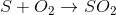
B)

C)
The conversion of fuel is only 80%
Fuel reacted = 5000(0.80) =4000 mol/h
Unreacted fuel = 1000 mol/h
Selectivity of CO2 to CO = 12
Amount of C reacted = 5000(0.80) (0.80) =3200 mol/h
Moles of C undergoing reaction 1 = 3200(12/13) = 2953.846 mol/h
Moles of C undergoing reaction 2 = 3200-2953.846 = 246.153 mol/h
Moles of H reacted = 5000(0.15) (0.80)
= 600 mol/h
Moles of S reacted = 5000(0.05) (0.80)
= 200 mol/h
According to stiochiometry O2 reacted = 2953.846(1) + 246.153(0.5) + 600(0.25) + 200(1) = 3426.9225 mol/h
Air contains 79% N2 and 21 % O2
O2 in feed = 35000(0.21) = 7350 mol/h
Product analysis based on stiochiometry
| Component | mol/h | mol% |
| Unreacted fuel | 1000 | 2.83 |
| CO2 | 2953.846(1)= 2953.846 | 8.37 |
| CO | 256.153 | 0.725 |
| H2O | 300 | 0.8502 |
| SO2 | 200 | 0.566 |
| N2 | 35000(0.79) = 27650 | 78.366 |
| O2 | 7350- 3426.9225= 3923.0775 | 11.11 |
| Total | 35283.076 | 100 |
D)
% excess air = % excess O2
O2 needed according to stiochiometry if all reactants reacted = (5000×0.8×(12/13) ×1)+ (5000×0.8×(1/13) ×0.5) + (5000×0.15×0.25) +(5000×0.05×1) = 4283.653 mol/h
O2 supplied = 7350 mol/h
% excess =( (7350-4283.653) /4283.653) (100) =
71.582 %
Please upvote if helpful
Related Solutions
a. 500 mol/h methane at 40°C was fed to a furnace and burned with 15% of...
A fuel gas containing methane and ethane is burned with air in a furnace, producing a...
A fuel gas containing methane and ethane is burned with air in a furnace, producing a...
Methane at 25°C is burned in a boiler furnace with 10.0% excess air. The air enters...
In a furnace, 76 lbs propane C3H8 are burned with 17.2% excess air for complete combustion....
100 m3 of a fuel oil are burned per hour with 20 percent excess air in...
A fired heater uses natural gas as fuel which is burned with 25% excess air. 90%...
100 mol n-Hexane (C6H14) is burned with excess air. An analysis of the product gas yields...
One kg of Methance (CH4) is burned with 200% theoretical air. What is the air fuel...
Pentane is burned with 80% excess air, 40% of the carbon that is oxidized goes to...
- Hi, how can I create a random guessing game in C++ with 2 players. I got...
- A long solenoid has 110 turns/cm and carries current i. An electron moves within the solenoid...
- Use the function from Q1 to find the sum of ints,n, from 1 to 2500, inclusive,...
- Normative Economics (Efficiency vs, Equity) there are two types of economic Efifficence Vs Equity why it...
- Any amount received or accrued by a taxpayer from an educational policy, which has been expended...
- Write a script that accepts a single character argument (command line parameter), then checks to see...
- Answer the following questions completely. Describe some of the differences between tariffs and quotas. What are...
 Amanda K answered 2 weeks ago
Amanda K answered 2 weeks ago