Question
In: Physics
A hydrogen atom, initially at rest, absorbs an ultraviolet photon with a wavelength of λ=177.9nm. Part...
A hydrogen atom, initially at rest, absorbs an ultraviolet photon with a wavelength of λ=177.9nm.
Part A:
What is the atom's final speed if it now emits an identical photon in a direction that is perpendicular to the direction of motion of the original photon?
Express your answer to three significant figures and include appropriate units.
Part B:
What is the atom's final speed if it now emits an identical photon in a direction that is opposite to the direction of motion of the original photon?
Express your answer to three significant figures and include appropriate units.
Solutions
Expert Solution
given
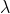 =
177.9 nm
=
177.9 nm
= 177.9 x 10-9 m
a )
using equation P = h / 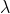
= 6.627 x 10-34 / 177.9 x 10-9
P = 3.7251 x 10-27 kg-m/sec
initial momentum Px = 3.7251 x 10-27 i
final momentum Py = 3.7251 x 10-27 j
change in momentum = 3.7251 x 10-27 i - 3.7251 x 10-27 j
magnitude = ( (3.7251 x 10-27 )2 + ( 3.7251 x 10-27 )2 )1/2
= 5.268 x 10-27 N-sec
here we have change in momentum is = m v
m v = 5.268 x 10-27
v = 5.268 x 10-27 / 1.67 x 10-27
so the atom's final speed if it now emits an identical photon in a direction
that is perpendicular to the direction of motion of the original photon is v = 3.1544 m/sec
b )
in opposite direction = ( m v- ( - mv ) )
= 2 m v
= 2 x 3.7251 x 10-27
then now
2 x 3.7251 x 10-27 = 1.67 x 10-27 x v
2 x 3.7251 = 1.67 x v
v = 2 x 3.7251 / 1.67
so the atom's final speed if it now emits an identical photon in a direction
that is opposite to the direction of motion of the original photon v = 4.4933 m/sec
Related Solutions
a hydrogen atom in the ground state absorbs a photon of light with a wavelength of...
A ground state hydrogen atom absorbs a photon of λ = 94.91 nm, exciting the electron...
A ground state hydrogen atom absorbs a photon of light having a wavelength of 93.03 nm....
what is the wavelength of the longest wavelength photon that a Ground state hydrogen atom can...
An electron in a Hydrogen atom originally at n=5 energy level absorbs a photon with a...
In Lyman series, one wavelength in the ultraviolet part of the hydrogen emission spectrum has wavelength...
A photon of wavelength 0.65037 nm strikes a free electron that is initially at rest. The...
An Electron initially in the n=2 state of a hydrogen atom is excited by a photon...
An electron jumps to the n=4 of a hydrogen atom after absorbing a photon of wavelength...
Compton Scattering.A photon of wavelength λcollides elastically with a free electron (initially at rest) of mass...
- Why is it significant that arginine vasopressin is expressed in squirrel monkeys and the amino acid...
- In the sigma-model of spontaneous symmetry breaking, we have degenerate vacuum states. But if we don't...
- Calculate the Kovats retention index for an unknown using the following retention times: 1.8 min for...
- the following selected financial statement information is for Stevens Company December 31 2017 2016 Changes in...
- A ball is thrown straight up from the edge of the roof of a building. A...
- Buffer A Buffer B Mass of NaC2H3O2 0.2449 2.449 Volume of buffer 100 100 M of...
- In the story "Superman and Me" by Sherman Alexie, The story opens by giving some reason...
 genius_generous answered 2 months ago
genius_generous answered 2 months ago