Question
In: Physics
electrons are ejected from a metallic surface with speeds ranging from 4.5 times ten to the...
electrons are ejected from a metallic surface with speeds ranging from 4.5 times ten to the -5 when light with a wavelength of 600 nm is used a) what is the work function of the surface? B) what is the cutoff frequency for the surface?
Solutions
Expert Solution
When photons with Energy E strike the metallic surface we have this realation
E= Work function + Kinetic Enegy
E=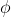 +
KE
+
KE
and energy E is
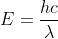
(where h is plank constant , c is speed of light and  is wavelength)
is wavelength)

E=3.315 10-19
J
10-19
J
and
Kinetic energy of ejected elcctron 
(where me is mass of electron and v is velocity of electron)
me= 9.1 x10-31 Kg
V= 4.5x10-5 m/s (unit not mentioned in problem is so taking m/s)
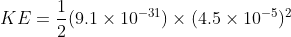
KE=92.13x10-41 J
Now
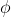 = E - KE
= E - KE
=(3.315 10-19
) -(92.13x10-41)
10-19
) -(92.13x10-41)
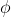 =3.315x
10-19 J (approx)
( as the value of KE is very small)
=3.315x
10-19 J (approx)
( as the value of KE is very small)
(B)
cutoff frequency is the frequency at which electron ejects.
Energy requried by elctron to get ejected = work function
(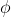 )
)
and we have
E =hf ( where f is frequency )
here E= 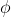
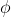 = hf
= hf
f= 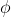 /h
/h
f=(3.315x 10-19)/(6.63x10-34)
f=5 10-15
Hz (ANS)
10-15
Hz (ANS)
Related Solutions
a) Electrons are ejected from a photoelectric surface with a maximum speed of 4.20 x 105...
When light of frequency 7.17x1014 Hz shines on the surface of sodium metal, electrons are ejected...
When light of frequency 4.96×1015 s-1 shines on the surface of a metal, electrons are ejected...
What is the maximum velocity of electrons ejected from a material by 90 nm photons, if...
The photoelectric effect is the emission of electrons from the surface of a conductor when light...
Light striking a metal surface causes electrons to be emitted from the metal via the...
- A student has two lenses, one of focal length 5.5 cm and the other with focal...
- Show and explain each step (neatly in complete sentences), indicate units, and specify the correct number...
- Amazon and Google have implemented cloud-based DBMS, what are some pros and cons? What are some...
- Terminology: 1.Thickness of internal coating of an analytical column (meaning, the purpose and the range of...
- im learning how to code and i hear what is important are data structures and algorithms....
- You have looked at the current financial statements for Reigle Homes, Co. The company has an...
- What is the circuit diagram of password based door lock system project by using interface 8088...
 genius_generous answered 2 months ago
genius_generous answered 2 months ago