Question
In: Chemistry
The heat of solution of LiCl is -37 kj/mol. a) draw a picture to illustrate the...
The heat of solution of LiCl is -37 kj/mol.
a) draw a picture to illustrate the main intermolecular force that is fromed in this process.
b) Explain why this process (dissolving LiCl in water) is exothermic.
Solutions
Expert Solution
Heat of solution is the energy required to dissolve a solute (here LiCl) into a solvent (here water). The negative sign implies that the energy is released when LiCl is dissolved in water. It means that the reaction is exothermic. First coming to part b) that why this reaction is exothermic?
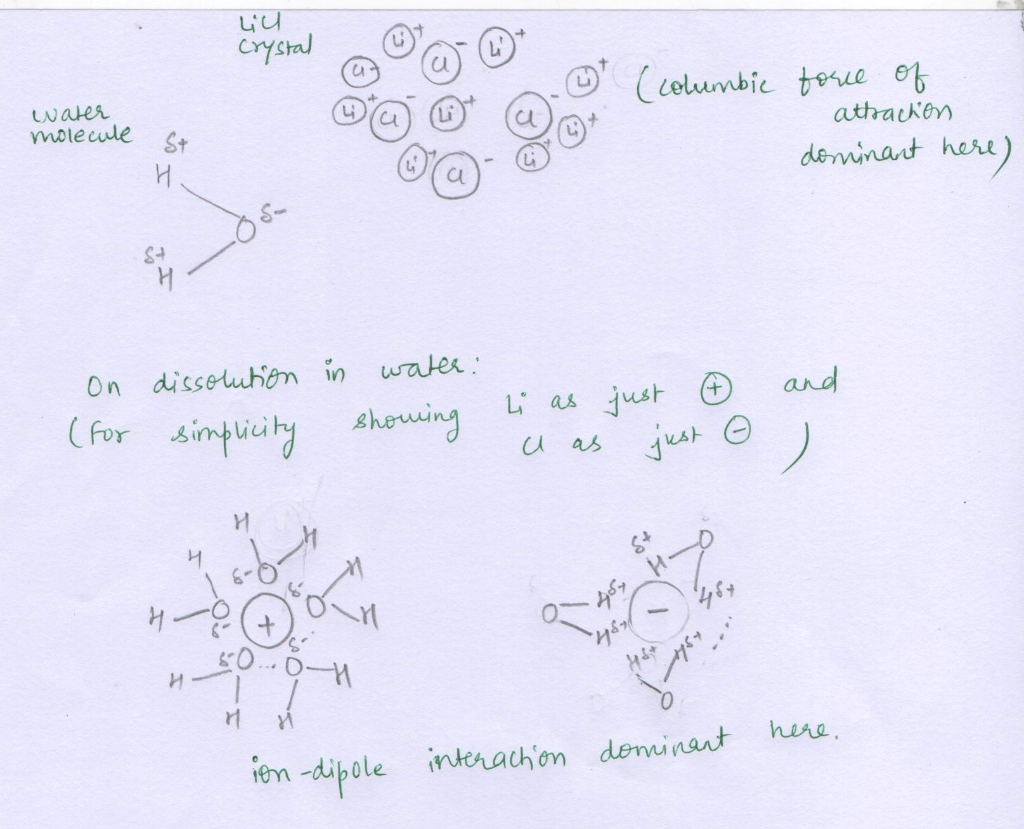
LiCl molecules are held together by Coulombic forces of attraction between Li+ cation and Cl- anion. The energy that binds the crystal of LiCl is the lattice energy. In other words the amount of energy required to place the ions ( Li+ and Cl-) very very far (infinity) from each other (that is breaking the crystal altogether!) is lattice energy.
When LiCl dissolves in water, the water molecules being polar (a dipole, having partial H + and partial O- in the same molecule), interact with both the ions Li+ and Cl- via ion-dipole interactions. These water molecules come in between the Li+ and Cl- ions and slowly cover these ions completely. The cation Li+, would be attracted to Oxygen atom of water and anion , Cl- would be attracted to Hydrogen atoms of water as shown in the attached diagram. Being very small in size, Li+ cation has high charge density and it is heavily hydrated by water molecules. So much so, that now Li+ and Cl- can't see each other at all! It has dissolved completely in water. So, the hydration energy for LiCl is greater than its lattice energy and overall energy is released in the process, leading to exothermic heat of solution.
a) The forces as mentioned above i.e. Coulombic forces of attraction and ion-dipole interactions are shown in the following diagram:
Related Solutions
1. For KNO3 the heat of solution is 23.8 kJ/mol and the lattice energy is -685.0...
The heat of solution of potassium acetate (KC2H3O2) in water is -15.30 kJ/mol. If 19.70 g...
Given that the heat of fusion of water is -6.02 kJ/mol, that the heat capacity of...
Given that the heat of fusion of water is -6.02 kJ/mol, that the heat capacity of...
Given that the heat of fusion of water is -6.02 kJ/mol, that the heat capacity of...
HCl: -164.4 kJ/mol NaOH: -469.6 kJ/mol NaCl: -407.1 kJ/mol H2O: -285.9 kJ/mol If 50 mL of...
The standard enthalpy of fusion of water is 6.01 kJ/ mol, the molar heat capacity at...
If the heat of combustion for a specific compound is -1270.0 kJ/mol and its molar mass...
the heat of fusion of ice is 6.00 kj/mol. what number of photons of wavelength =...
Answer the following questions: a). The heat of fusion of ice is 6.00 kJ/mol. Find the...
- c++ 1 city.h #pragma once #ifndef CITY_H #define CITY_H #include<string> #include<iostream> #include<utility> using namespace std; class...
- 1. Complete the balance sheet for the business for 2016 and 2017. 2. In which year...
- 1. What is oxidation? How do you know if a molecule/atom has undergone oxidation. 2. Describe the...
- write a simple python program that takes in 6-50 random numbers the user types in and...
- What are “perceptual committees”? Is there ever an incident where perceptual committees fail? If so, why?...
- Please explain answer a little bit I'm familiar with these all topics..just give hint with your...
- Calculate the pH of a buffer solution after the addition of 15.00 mL of 0.100 M...
 queen_honey_blossom answered 1 month ago
queen_honey_blossom answered 1 month ago