Question
In: Physics
A glass flask whose volume is 1000cm3 at a temperature of 1.00?C is completely filled with...
A glass flask whose volume is 1000cm3 at a temperature of 1.00?C is completely filled with mercury at the same temperature. When the flask and mercury are warmed together to a temperature of 52.0?C , a volume of 8.35cm3 of mercury overflows the flask. If the coefficient of volume expansion of mercury is ?Hg = 1.80
Solutions
Expert Solution
Heated volume of Hg = Original volume of Hg{1 + 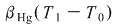 }
}
Orginal volume of Hg = 1000 cm^3, 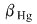 =1.80 x 10^-4,
=1.80 x 10^-4,
T1-T0 = change in temperature = 52-1= 51 C
Heated volume of Hg= 1000{ 1+ 1.80x10^-4 x 51}
heated volume of Hg= 1009.18 cubic centimeter
Heated volume of glass= Heated volume of Hg- amount of mercury overflowing
Heated volume of glass = 1009.18-8.35= 1000.83 cubic centimeter
Heated volume of glass=Original volume of glass { 1 + 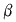 glass(T1-T0)}
glass(T1-T0)}
1000.83 = 1000{ 1+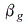 (51)}
(51)}
1.00083 = 1 + beta(51)
0.00083 = 51*beta
coefficient of expansion of glass = 0.00083/51
= 1.627 x 10^-5 /K
Related Solutions
A glass flask whose volume is 1000 cm3 at a temperature of 0.100 ∘C is completely...
Wine bottles are never completely filled: a small volume of air is left in the glass...
A 1.00 L flask is filled with 1.45 g of argon at 25 ∘C. A sample...
A 1.00 L flask is filled with 1.20 g of argon at 25 ∘C. A sample...
A 1.00 L flask is filled with 1.45 g of argon at 25 ∘C. A sample...
Finding the volume of a flask. A student obtained a clean dry glass-stoppered flask.
A flask has a volume of 159.6 mL . When filled with air at a pressure...
a study was made of the effect of the temperature on the volume of 1.00 g...
A balloon filled with helium has a volume of 30.0 L at a pressure of 75 kPa and a temperature of 35.0°C.
A helium-filled balloon has a volume of 0.010 m3. The temperature in the room and in...
- What defines a “rat race”? What makes consumption into a “rat race”?
- Diversity Does the growing diversity in the United States pose special challenges for law enforcement and...
- A football player kicks the ball so that it will have a "hang time" (time of...
- The purpose of this exercise is to provide the Marketing department with a method to sort...
- A student has two lenses, one of focal length 5.5 cm and the other with focal...
- Show and explain each step (neatly in complete sentences), indicate units, and specify the correct number...
- Amazon and Google have implemented cloud-based DBMS, what are some pros and cons? What are some...
 genius_generous answered 3 months ago
genius_generous answered 3 months ago