Question
In: Chemistry
1.) Chlorofluorocarbons (CFCs) were introduced as refrigerants in the 1930s, replacing other gases that were toxic...
1.) Chlorofluorocarbons (CFCs) were introduced as refrigerants in the 1930s, replacing other gases that were toxic and corrosive. How do the molecular shape and polarity of CFCs contribute to the many desirable qualities they have as refrigerants? Given that they are such desirable refrigerants, why are they banned? What chemistry is involved? How has their molecular shape and polarity had harmful effects on the atmosphere?
2. Chemically, how does soap work to remove grease? Give some other related examples.
Solutions
Expert Solution
1.Carbon in CFCs are arranged in tetrahedral symmetry.because of large difference in atomic size between hydrogen and flurine it deviate from perfect tetrahedral symmetry. they are volatile compounds,but less than their parent alkanes. the decreased polarity attributed to the molecular polarity induced by the halides. the CFCs have high boiling point because chloride is more polarizable than fluoride. because of good polarity CFCs are good solvents and their boiling point make them suitable as refrigerant.
Eventhough they are suitable refrigerant they are banned because they harmful to ozone in the atmosphere. the most important chemistry behind this is CFCs cause the photo induced splitting of c-cl bond.
CCl3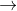 CCl2+Cl-
CCl2+Cl-
the chlorine atom behaves different from ordinary chlorine molecule. it has long lived in upper atmosphere and catalyses the splitting of ozone in to o2. the depletion of ozone causes the arrival more solar radiation towards earth surface. so that the temperature on the earth increases this leads to green house effect.this is the reason for CFCs considered as a harmful substance and it is banned.
2.The working principle of cleansing action of soap is as follows. soap is made up of an emulsifier. it containes at one end hydrophilic part[water loving] and at the another end hydrophobic part[oil loving part]. the hydrophobic part attached to the grease or oil part in the cloths and the hydrophilic part attached to the water part. while we rubbing the cloths by friction the water grabs the oil loving end along with the grease its attached to, and carried off.
another examples of this types are detergent , shampoos, shaving creams etc.
Related Solutions
How many grams of Cl are in 38.8 g of each sample of chlorofluorocarbons (CFCs)? 1....
1 mole of H2 and 0.5 mole of O2 gases at 298K are introduced into an...
Annual emissions of chlorine in CFCs were less than 1% by mass of the chlorine emitted...
1) Compare the toxic effects for “arrowhead poison” versus “nerve gases”. 2) Compare the effects of...
You were introduced to some common administrative commands in this chapter. However, there are other utilities...
1. Students were introduced to events that were independentand those events that were not independent within...
1. What problems are associated with oxygen or other gases that are dissolved in molten steel? 2. What...
1. Activity Based Costing Duncan Company recently introduced a markers to complement their other product, pencils....
1. Identify four new materials in modern automotive industries that were introduced in the last twenty...
Consider the six theoretical orientations you were briefly introduced to in the Unit 1 video. Which...
- im learning how to code and i hear what is important are data structures and algorithms....
- You have looked at the current financial statements for Reigle Homes, Co. The company has an...
- What is the circuit diagram of password based door lock system project by using interface 8088...
- java programming You will be given two interfaces and two abstract classes, FileTextReader, FileTextWriter, AbstractFileMonitor, and...
- Why is it significant that arginine vasopressin is expressed in squirrel monkeys and the amino acid...
- In the sigma-model of spontaneous symmetry breaking, we have degenerate vacuum states. But if we don't...
- Calculate the Kovats retention index for an unknown using the following retention times: 1.8 min for...
 queen_honey_blossom answered 3 months ago
queen_honey_blossom answered 3 months ago