Question
In: Chemistry
Let’s compare two monoprotic acids: one strong (nitric, HNO3, pKa = -1.60) and one weak (acetic,...
Let’s compare two monoprotic acids: one strong (nitric, HNO3, pKa = -1.60) and one weak (acetic, CH3COOH, pKa = 4.79).
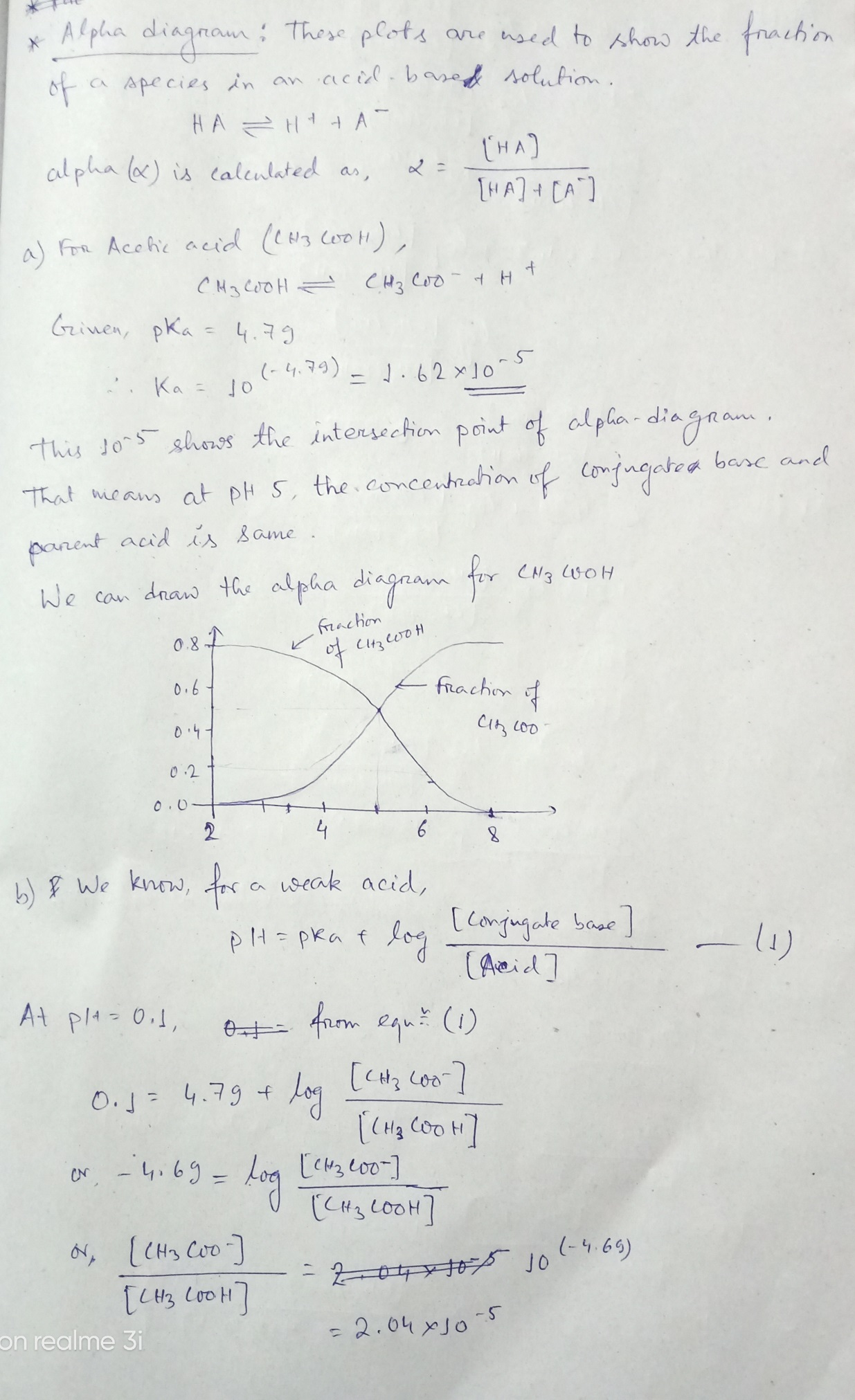
a. [4 points] Sketch an alpha diagram for each of the acids.
b. [6 points] Use your alpha diagram to predict the percentage of undissociated acid for each at the following pH values: 0.1, 3, 4.3, 6, and 13. After estimating the fractions with your diagram, check your values by calculating the exact value.
c. [4 points] Explain the difference in pKa values (and the percentage dissociated in parts a and b) for nitric acid and acetic acid. In your explanation, include and underline the following terms: pH, H+ concentration, strong/weak acid, and Le Chatelier’s Principle.
Solutions
Related Solutions
If pKa of weak acids are the following: pKa acetic acid = 4.75 pKa propionic acid...
If
pKa of weak acids are the following: pKa acetic acid = 4.75 pKa
propionic acid = 4.87 pKa acitric acid = 5.40 pKa of benzoic acid =
4.19
What is the order of acids with increasing acidity Determine
the pH of the following
A. 100 mL of benzoic acid solution 0.03M is mixed with 200 ml
water
B. 50 mL of sodium hydroxide solution 0.05 M is mived with 200
ml water
Acetic acid is a monoprotic weak acid with a pKa of 4.74. (Ka = 1.8 x...
Acetic acid is a monoprotic weak acid with a pKa of 4.74. (Ka =
1.8 x 10-5)
(a) What is the pH of 5 mL of a 5.0% solution?
(b) What is the pH of the solution if you now add 45 ml of water to
solution (a)?
How will the equivalence point volumes differ if you titrate the
two solutions ?
1: classify the below list as Strong acids Weak acids Strong bases Weak bases HNO2...
1: classify the below list as Strong acids Weak
acids Strong bases Weak bases
HNO2
HCl
LiOH
HClO4
Ca(OH)2
NH3
HCN
Na3PO4
2:classify as forward or
reverse favored reaction
CH3COOH(a q)+H2O(l)⇌CH3COO−(aq)+H3O+(aq)
H2SO4(l)+H2O(l)⇌H3O+(aq)+HSO4−(aq)
HCN(l)+H2O(l)⇌CN−(aq)+H3O+(aq)
HCl(l)+OH−(aq)⇌H2O(l)+Cl−(aq)
Make a list of the anions of the six strong monoprotic acids discussed in lecture. These...
Make a list of the anions of the six strong monoprotic acids
discussed in lecture. These anions are said to have no effect on
the pH of an aqueous solution. The same is true for solutions of
strong bases - their conjugate acids (which are Group 1 or Group 2
cations) are very weak acids, and therefore, have virtually no
effect on the pH of the solution.
4. Make a list Group 1 and Group 2 cations, and note that...
The pKa of formic acid is 3.74, a monoprotic weak acid. A 1.0 L sample of...
The pKa of formic acid is 3.74, a monoprotic weak acid. A 1.0 L
sample of a buffer with a pH of 4.5 is combined with .09 mol of
NaOH. What is the new pH of the solution? The ratio of HCOO- to H+
is .86 M to .15 M.
Consider a monoprotic weak acid (HA) that is titrated with a strong base. What is the...
Consider a monoprotic weak acid (HA) that is titrated with a strong base. What is the relationship between the strength of the weak acid and the pH of the solution at the equivalence point?
There is no relationship between the strength of the acid and the pH at the equivalence point.
The pH at the equivalence point is always 7 in an acid base titration.
The weaker the acid, the higher the pH at the equivalence point.
The stronger the...
Consider the following three acids: a. Nitric acid trihydrate ( HNO3 . 3H2O , a monoprotic...
Consider the following three acids:
a. Nitric acid trihydrate ( HNO3 . 3H2O ,
a monoprotic acid with molar mass = 117.07g/mol )
b. Oxalic acid ( H2C2O4, a
diprotic acid with molar mass = 90.04 g/mol)
c. Phosphoric acid ( H3PO4, a triprotic
acid with molar mass = 98.00 g/mol)
A 1.50 g sample of one of the above acids is dissolved in enough
water to make a 125.ml acid solution. If 26.40 ml of 1.26 M NaOH is...
find the ph of each mixture of acids 0.020m HNO3 and 0.225 Mbr 0.095m in acetic...
find the ph of each mixture of acids
0.020m HNO3 and 0.225 Mbr
0.095m in acetic acid and 0.075.m in H2SO4
0.0500 M in HCL04 and 0.02 M HBR
Unlike strong acids and bases that ionize completely in solution, weak acids or bases partially ionize....
Unlike strong acids and bases that ionize completely in
solution, weak acids or bases partially ionize. The tendency to
ionize (i.e., the ionic strength) of a weak acid or base can be
quantified in several ways including
Ka or Kb,
pKa or pKb,
and percent ionization.
Part A
Pyridine is a weak base that is used in the manufacture of
pesticides and plastic resins. It is also a component of cigarette
smoke. Pyridine ionizes in water as follows:
C5H5N+H2O⇌C5H5NH++OH−
The...
Unlike strong acids and bases that ionize completely in solution, weak acids or bases partially ionize....
Unlike strong acids and bases that ionize completely in
solution, weak acids or bases partially ionize. The tendency of a
weak acid or base to ionize can be quantified in several ways
including
Ka or Kb,
pKa or pKb,
and percent ionization*.
*This assumes that species of equal concentrations are being
compared as percent ionization is affected by concentration.
Part A
Pyridine is a weak base that is used in the manufacture of
pesticides and plastic resins. It is also...
ADVERTISEMENT
ADVERTISEMENT
Latest Questions
- Why is it significant that arginine vasopressin is expressed in squirrel monkeys and the amino acid...
- In the sigma-model of spontaneous symmetry breaking, we have degenerate vacuum states. But if we don't...
- Calculate the Kovats retention index for an unknown using the following retention times: 1.8 min for...
- the following selected financial statement information is for Stevens Company December 31 2017 2016 Changes in...
- A ball is thrown straight up from the edge of the roof of a building. A...
- Buffer A Buffer B Mass of NaC2H3O2 0.2449 2.449 Volume of buffer 100 100 M of...
- In the story "Superman and Me" by Sherman Alexie, The story opens by giving some reason...
ADVERTISEMENT


 queen_honey_blossom answered 3 months ago
queen_honey_blossom answered 3 months ago