Question
In: Other
Determine ionic strength, I and mean ionic activity coefficient, γ± at 298 K for 0.025 molal...
- Determine ionic strength, I and mean ionic activity coefficient, γ± at 298 K for
- 0.025 molal K2SO4
- 0.01 molal Li2CO3
- 0.01 molal Ca2(PO4)2
- Estimate the solubility of
- MgF2 (Ksp=6.4x10-9) in an aqueous solution with I=0.003 molal.
- BaSO4 (Ksp=1.08x10-10) in an aqueous solution with I=0.001 molal.
Solutions
Expert Solution
Ionic strength (I) is given by the formula,
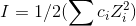
ci denotes the concentration of the ion "i" and Z denotes the valency of the concerned ion.
(i)
0.025 molal K2SO4
K2SO4 dissociates to giving 2 K+ ions and 1 SO42- ions.
When we gave 0.025 molal of K2SO4, we get 0.05 molal of K+ ions a 0.25 molal of SO42- ions.
Therefore, For K+ ions, c = 0.05 and valency is the charge on the ion and is equal to +1
For SO42- ions, c = 0.025 and valency is -2
Therefore, ionic strength:
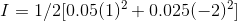
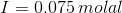
(ii)
0.01 Li2CO3
1 mole Li2CO3 dissociates to give 2 moles of Li+ and 1 mole of CO32-
Therefore when we have 0.01 molal of Li2CO3, we get 0.02 molal of Li+ and 0.01 molal of CO32-
Valency of Li+= 1 and Valency of CO32-= -2
Therefore,
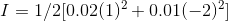
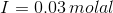
(iii)
0.01 molal Ca3(PO4)2
1 mole of Ca3(PO4)2 dissociates to give 3 moles of Ca2+ ion and 2 moles of PO43- ion.
Therefore, 0.01 molal of Ca3(PO4)2 gives 0.03 molal of Ca2+ and 0.02 molal of PO43-.
Valency of Ca2+ = +2 and Valency of PO43-= -3
Therefore, Ionic strength :
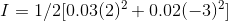
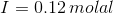
Solubilities:
(i)
We need to calculate the solubility of MgF2 in a solution of MgF2 with ionic strength of 0.003 molal
MgF2 dissociates into Mg2+ and 2 F-. Therefore let the concentration of Mg2+ be X molal and therefore the concentration of F- will be 2X molal.
Valency of Mg2+ = +2 and the Valency of F- = -1
Given: Ksp = 6.4 x 10-9
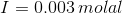
On substituting the values of concentration and valency in the formula for Ionic strength, we get:
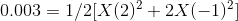
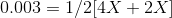
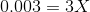
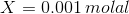
Therefore, concentration of the Mg2+ is 0.001 molal and the concentration of the F- is 0.002 molal
Ksp is given by the formula,
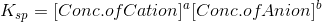
where a is the number of molecules of cation formed and b is the number of molecules of anion formed.
From the given sample of MgF2, Let X be the molal of MgF2 that actually dissociates, therefore, concentration of Mg2+ formed is X and that of F- is 2X.
a for Mg2+ is 1 and b for F- is 2 as MgF2 dissociates into 1 molecule of Mg2+ and 2 molecule of F-
As, the solution already contains some dissolved MgF2, we will have to consider this concentration as well.
as calculated above, the total Concentration of Mg2+= X+0.001
The total concentration of F-= 2X+0.002
Therefore,
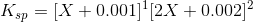
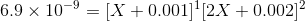
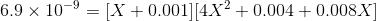
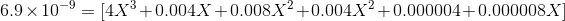
Ksp is in the order of magnitde of 10-9 and therefore its safe to say X will be also in a similar order of magnitude because of which we can ignore powers of 2 and 3 in X as well as the last 2 terms, therefore we get:
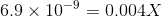
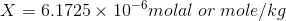
X is the desired solubility of the compound
(ii)
BaSO4
BaSO4 dissociates into Ba2+ and SO42-.
The given solution has an ionic strength of 0.001 molal
let the concentration of Ba2+ be X then the concentration of the SO42+ will also be X.
Valency of Ba2+ and SO42- is (+2) and (-2) respectively.
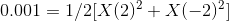
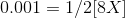
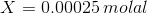
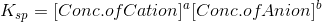
a =1 and b=1 as only 1 unit of Ba2+ and SO42- is formed.
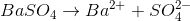
Therefore, if we have X is the concentration of BaSO4, the concentration of Ba2+ and SO42- based on stoichiometry is X and X molal
Total Concentration of Ba2+= X + 0.00025 molal
Total Concentration of SO42-= X + 0.00025 molal
Ksp= 1.08 x 10-10
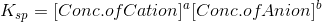
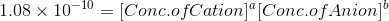
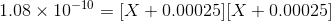
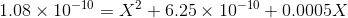
Ignoring X2 and the second term, we get :
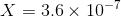
X is the required solubility.
Related Solutions
Explain why: Ionic strength increase then activity coefficient decrease Ionic charge increase then activity coefficient increase...
Explain why (Simple explanation) Ionic strength increase then activity coefficient decrease Ionic charge increase then activity...
21. determine the activity coefficient given the molal concentration of ZnBr2 is 0.01 mol/kg.
8. Determine the ionic strength of the solution and the activity coefficients for each ion in...
why is it important to be able report the mean activity coefficient of an ionic solute?
Using the definition of Ka and the Debeye-Huckel expression for the mean ionic activity coefficient, calculate...
calculate the mean ionic activity of a 0.0350 m Na3PO4 solution for which the mean activity...
The strength coefficient (K) and the work hardening exponent (n) for a stainless steel were determined...
Use the values found in Thermodynamic Properties to determine ΔS°, ΔH° and ΔG° at 298 K...
Consider the following reaction at constant P. Determine the value of ΔSsurroundings at 298 K? Predict...
- What defines a “rat race”? What makes consumption into a “rat race”?
- Diversity Does the growing diversity in the United States pose special challenges for law enforcement and...
- A football player kicks the ball so that it will have a "hang time" (time of...
- The purpose of this exercise is to provide the Marketing department with a method to sort...
- A student has two lenses, one of focal length 5.5 cm and the other with focal...
- Show and explain each step (neatly in complete sentences), indicate units, and specify the correct number...
- Amazon and Google have implemented cloud-based DBMS, what are some pros and cons? What are some...
 Amanda K answered 2 months ago
Amanda K answered 2 months ago