Question
In: Chemistry
The formate ion, HCO2-, is formed when formic acid dissolves in water.
The formate ion, HCO2-, is formed when formic acid dissolves in water. A number of possible resonance structures for this ion are shown.
Which of these structures are valid and which are invalid?
Drag each item to the appropriate bin.
Solutions
Expert Solution
Concepts and reason
Resonance structures are Lewis structures that are obtained due to the motion of lone pair or pi-electron cloud in the structure.
These structures are electronically equivalent and indicated by double-headed arrow \((\leftrightarrow)\)
Fundamentals
Resonance structures : Resonance structure are structures which are indicating the same molecule but differ in their electrinical geometry. Let \(\mathrm{A}, \mathrm{B},\) and \(\mathrm{C}\) are resonance structures, indicated as follows. \(\mathrm{A} \leftrightarrow \mathrm{B} \leftrightarrow \mathrm{C}\)
Charge on every canonical structure (resonance structure) should be equal.
Given ion is formate ion \(\left(\mathrm{HCO}_{2}^{-}\right)\) Given that this formate ion produced by dissolving the formic acid in water. Complete reaction is as follows:
\(\mathrm{HCOOH}+\mathrm{H}_{2} \mathrm{O} \longrightarrow \mathrm{HCOO}^{-}+\mathrm{H}_{3} \mathrm{O}^{+}\)
Lewis structure of the formate ion is shown below.
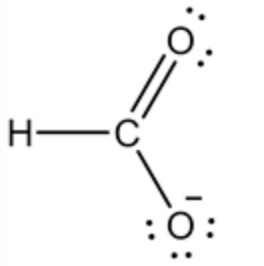
Formate ion is produced by dissolving the formic acid in the water, this reaction has been shown. And the Lewis dot structure has been given for the formate ion.
Formate ion structure is shown below:

The resonance structure for this formate ion is as follows:
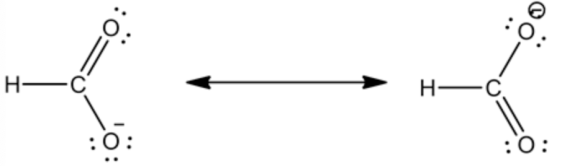
Therefore, only two possible resonance structures are there for formate ion.
Resonance structures have been drawn for the formate ion, which is produced during the dissolving process of formic acid in water.
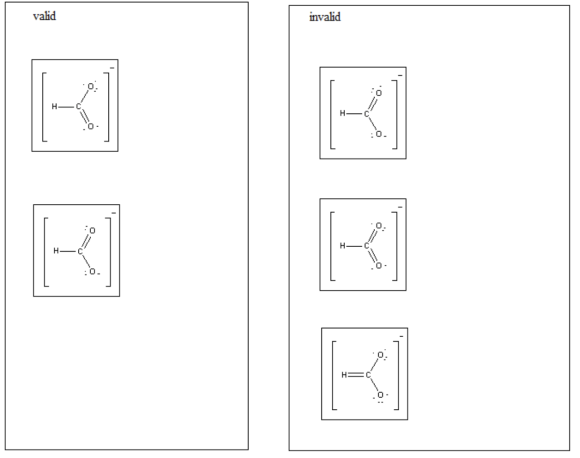
Related Solutions
A.) What ions are formed when KCN dissolves in water? Identify the ion in KCN which...
A chemist is working with a formic acid � formate ion buffer system. HCHO2(aq) + H2O(l)...
(19) Ammonium formate, NH4(HCOO), dissolves in water to give ammonium ions and formate ions. These ions...
(a) What is the pH of 0.89 M potassium formate, HCOOK? (The Ka of formic acid...
1. Use the Henderson-Hasselbalch equation to determine the ratio of base/acid in a formic acid-formate buffer...
Formic acid,HCHO2 , is used to make methyl formate (a fumigant for dried fruit) and ethyl...
With the same initial solution of 0.75 moles formate and 0.85 moles formic acid to make...
the Al(H2O)63+ ion is formed when an Al3+ ion acting as a Lewis acid picks up...
A.You have 1 M solutions of formic acid and sodium formate. Calculate the volume of sodium...
2.0 mL of 0.5M HCl was added to 50 mL of 0.5M Formate-formic acid buffer at...
- What is the circuit diagram of password based door lock system project by using interface 8088...
- java programming You will be given two interfaces and two abstract classes, FileTextReader, FileTextWriter, AbstractFileMonitor, and...
- Why is it significant that arginine vasopressin is expressed in squirrel monkeys and the amino acid...
- In the sigma-model of spontaneous symmetry breaking, we have degenerate vacuum states. But if we don't...
- Calculate the Kovats retention index for an unknown using the following retention times: 1.8 min for...
- the following selected financial statement information is for Stevens Company December 31 2017 2016 Changes in...
- A ball is thrown straight up from the edge of the roof of a building. A...
 Dr. OWL answered 5 years ago
Dr. OWL answered 5 years ago