Question
In: Chemistry
Enzymes: what is a transition state complex?
Enzymes:
what is a transition state complex?
Solutions
Expert Solution
enzymes which are commonly known as Biocatalyst, present inside the cell. They help in catalysing the chemical reactions occuring in cell.. All enzymes are proteins or we can say proteinaceous in nature. They are important for life and carried out various functions like digestion, metabolism etc.
Just like catalyst in chemical reactions, enzyme increases the rate of reaction without getting coonsumed. In graph we can see both reactants and products of chemical reactions posses a characteristic amount of energy. The peak with maximum potential energy is Transition state .enzymes lowers the height of peak and reduces the energy that reactants must supply to form product
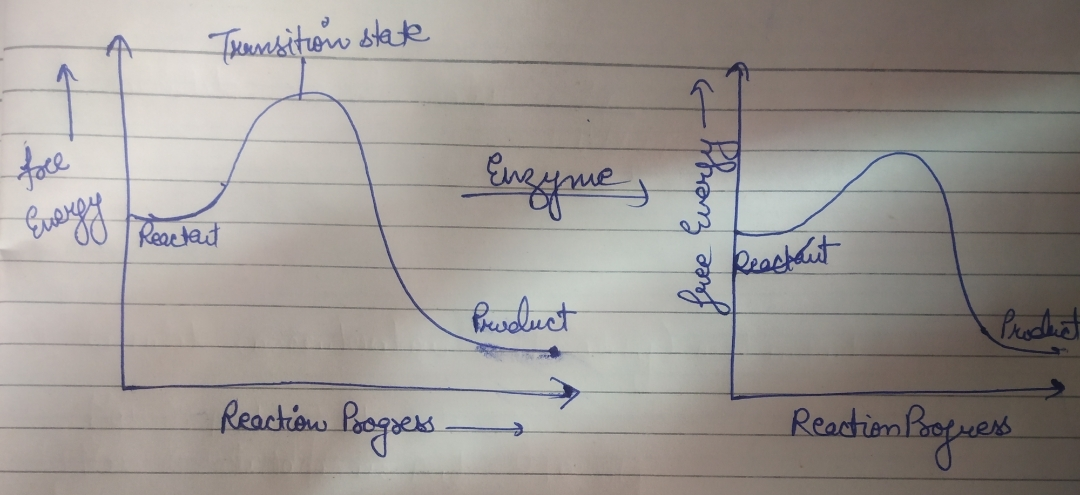
during this mechanism the enzyme bind to reactant with its active site from one side lowers its energy and further make modifications to convert it into product

Related Solutions
1. Is it reasonable to say that enzymes lower the energy of the transition state for...
Describe transition state theory and how it applies to enzyme catalysis. Why is a transition-state analog...
In a state machine diagram with composite states, what is the meaning of a transition that...
In a state machine diagram with composite states, what is the meaning of a transition that...
In what respects does a UML state diagram differ from a state transition diagram?
Chemical reactions are often described using a three state model: Reactants→Transition State→ProductsReactants→Transition State→Products In most cases...
Enzymes are kinetically important for many of the complex reactions for plant and animal life to...
What role does the transition state play in the decrease in activation energy that results with...
For the hydrogen atom, the transition from the 2p state to the 1s state is accompanied...
Enzymes can be thought as being catalytic receptors that bind transition states more strongly than the...
- Dorchester Company, on March 1, 2019 has a beginning Work in Process inventory of zero. All...
- This is to be done in C++. Preferably using basic functions. Write a loop that reads...
- Enzymes: what is a transition state complex?
- John’s Specialty Store uses a periodic inventory system. The following are some inventory transactions for the...
- What is the objective of this assignment? Briefly explain the Doppler Effect. Set both the listener...
- python- please finish the three programs, where one flips an image horizonally, then vertically, and one...
- A cat rides a merry go round turning with uniform circular motion. At time t1=2.00s, the...
 queen_honey_blossom answered 1 hour ago
queen_honey_blossom answered 1 hour ago